Sulfate-reducing bacteria (SRB) can enhance the uptake of silver-containing nanoparticles by a wetland plant†
Abstract
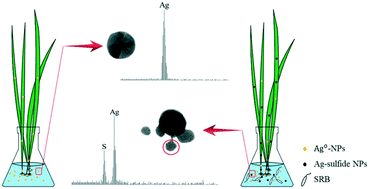
The presence of sulfate-reducing bacteria (SRB) can reduce the bioavailability of toxic metal ions (e.g., Ag+) to plants via mediating the formation of metal sulfide precipitates; however, it remains largely elusive if SRB can also affect the phyto-uptake of metal nanoparticles (e.g., Ag0-NPs). In the current study, the bioavailability of Ag0-NPs to a model wetland plant, Scirpus triqueter, was investigated in the presence/absence of SRB. Comparative experiments were conducted using 0.01–10 mg L−1 Ag0-NPs and silver ions. In addition to quantifying the total dissolved Ag concentrations, we analyzed the average sizes and particle concentrations of Ag-containing NPs (Ag-NPs) in plant tissues, including both roots and stems, after the designated treatments. The results show that although the presence of SRB can reduce the uptake of total Ag by 37% during the exposure of the plant to Ag ions, it can significantly enhance the uptake of total Ag during exposure of the plant to Ag0-NPs, likely by transforming Ag0-NPs into Ag-sulfide NPs with smaller particle sizes. Transmission electron microscopy data revealed that biogenic secondary Ag-sulfide particles smaller than 10 nm in size form in the vicinity of pristine Ag0-NPs. These NPs are likely generated from the parent Ag0-NPs via a dissolution–diffusion–sulfidation process. Moreover, the phyto-uptake of Ag0-NPs of various sizes (i.e., 20, 40 and 80 nm) in the presence/absence of SRB also confirmed a size dependent pattern, with more silver identified in the plant exposed to smaller Ag-NPs. The combined results suggest that the enhanced bioavailability of Ag-NPs to Scirpus triqueter in the presence of SRB is mainly attributed to the formation of secondary biogenic NPs with minute size. This result points to the importance of complex, coupled interactions between aqueous solutions, bacteria, plants, and labile NPs.


 Please wait while we load your content...
Please wait while we load your content...