Delineating the influence of picolinic acid on Eu(iii) sorption by γ-alumina†
Abstract
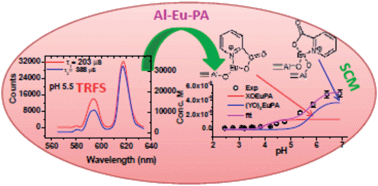
The present study aims at understanding the sorption mechanism of Eu(III) by γ-alumina in the presence of picolinic acid (PA), a decontaminating agent used in the nuclear industry, through batch sorption studies, spectroscopy and surface complexation modeling. PA is weakly sorbed by γ-alumina, with the sorption increasing with pH up to 4.5 and decreasing with further increase in pH. Attenuated Total Reflection-Fourier Transform Infrared Spectroscopy (ATR-FTIR) measurements indicate that PA forms an inner sphere surface complex on γ-alumina. The presence of PA does not affect Eu(III) sorption by γ-alumina at low [Eu(III)], while it drastically reduces Eu(III) sorption at high [Eu(III)]. Similar Eu(III) sorption profiles with different addition sequences of Eu(III) and PA suggest identical Eu(III) surface species for Eu(III) sorption on γ-alumina in the presence of PA which has been validated by time resolved fluorescence spectroscopy (TRFS). TRFS measurements of Eu(III) sorbed on γ-alumina in the absence (binary systems) and presence of PA (ternary systems) exhibited two components 1 & 2. The lifetime value of component 1 in ternary systems is enhanced compared to that in binary systems signifying the formation of new surface species containing Eu(III), PA and the γ-alumina surface whereas the similarity of component 2 in both the binary and ternary systems reveals an almost identical coordination environment of Eu(III) in the two types of system. Using the spectroscopic information obtained from TRFS, Eu(III) sorption, at high [Eu(III)], in ternary systems has been successfully modeled by considering Eu(III) bridged PA surface species at both low and high affinity sites of γ-alumina. At low [Eu(III)] both PA and Eu(III) bridged ternary surface complexes only at high affinity sites of γ-alumina could describe the Eu(III) sorption adequately.


 Please wait while we load your content...
Please wait while we load your content...