Ce(OH)2Cl and lanthanide-substituted variants as precursors to redox-active CeO2 materials†
Abstract
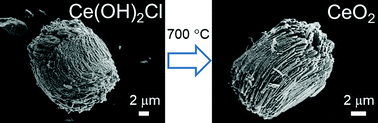
The cerium(III) hydroxide chloride Ce(OH)2Cl crystallises directly as a polycrystalline powder from a solution of CeCl3·7H2O in poly(ethylene) glycol (Mn = 400) heated at 240 °C and is found to be isostructural with La(OH)2Cl, as determined from high-resolution synchrotron powder X-ray diffraction (P21/m, a = 6.2868(2) Å, b = 3.94950(3) Å, c = 6.8740(3) Å, β = 113.5120(5)°). Replacement of a proportion of the cerium chloride in synthesis by a second lanthanide chloride yields a set of materials Ce1−xLnx(OH)2Cl for Ln = La, Pr, Gd, Tb. For La the maximum value of x is 0.2, with an isotropic expansion of the unit cell, but for the other lanthanides a wider composition range is possible, and the lattice parameters show an isotropic contraction with increasing x. Thermal decomposition of the hydroxide chlorides at 700 °C yields mixed-oxides Ce1−xLnxO2−δ that all have cubic fluorite structures with either expanded (Ln = La, Gd) or contracted (Ln = Pr, Tb) unit cells compared to CeO2. Scanning electron microscopy shows a shape memory effect in crystal morphology upon decomposition, with clusters of anisotropic sub-micron crystallites being seen in the precursor and oxide products. The Pr- and Tb-substituted oxides contain the substituent in a mixture of +3 and +4 oxidation states, as seen by X-ray absorption near edge structure spectroscopy at the lanthanide LIII edges. The mixed oxide materials are examined using temperature programmed reduction in 10%H2 in N2, which reveals redox properties suitable for heterogeneous catalysis, with the Pr-substituted materials showing the greatest reducibility at lower temperature.


 Please wait while we load your content...
Please wait while we load your content...