Quadruple bonding of bare group-13 atoms in transition metal complexes†
Abstract
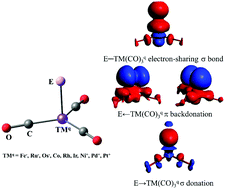
Density functional theory calculations at the M06-D3/def2-TZVPPD level of the group-13 anion complexes EFe(CO)3− (E = B–Tl) and the isoelectronic neutral and charged boron adducts BTM(CO)3q (TMq = Fe−, Ru−, Os−, Co, Rh, Ir, Ni+, Pd+, Pt+) give tetrahedral (C3v) geometries in the 1A1 electronic ground state as equilibrium structures. The analysis of the bonding situation with the energy decomposition analysis in combination with natural orbital for chemical valence method suggests that the E–TM(CO)3q bonds possess four bonding components: (a) one strong electron-sharing σ bond E–TM(CO)3q; (b) two π backdonations E⇇TM(CO)3q and (c) one weak σ donation E→TM(CO)3q. The relative strength of the four bonding components depends on the charge of the system, the transition metal TM and the group-13 atom E. The σ donation E→TM(CO)3q is in all systems rather weak while the associated charge migration is not negligible. A similar situation of the bonding of terminal group-13 atoms Ga and In is found in Ga–TM(GaCp)4+ and E–Pt(PMe3)3+ (TM = Ni, Pd, Pt; E = Ga, In), which are model compounds for the stable complexes GaTM(GaCp*)4+ (TM = Ni, Pt) and InPt(PPh3)3+. The quadruple bonds E→TML2 are hybrids of electron-sharing and dative bonds.


 Please wait while we load your content...
Please wait while we load your content...