Synthesis, structure and reactivity of NO+, NO˙ and NO− pincer PCN-Rh complexes†
Abstract
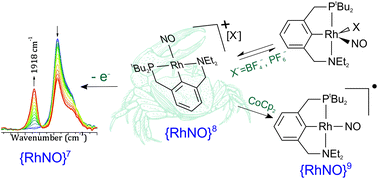
Synthesis of a pincer-type linear nitrosyl complex [Rh(PtBu2CNEt2)(NO)]+ (3+) is described. The product and all intermediates involved were fully characterized by FTIR, NMR, cyclic voltammetry and X-ray crystallography. Attempts at obtaining (3+) from its chlorinated precursor Rh(PCN)(NO)Cl (2) revealed that a relative stabilization of this complex ion is introduced by the BArF− counteranion, as other counteranions—PF6−, BF4− and triflate—proved to coordinate to the metal center. Redox reactivity both of (3+) and of that of its five-coordinate derivatives (2) and [Rh(PCN)(NO)(CH3CN)]+ (4+) was found to distinguish itself from analogous PCP complexes due to a relative stabilization of higher oxidation states. Oxidation of these three complexes was studied by FTIR spectroelectrochemistry. Reduction of complex (3+) to yield a short-lived {RhNO}9 species [Rh(PCN)(NO)]˙ (3˙) was also carried out. Complex (3˙) was proved able to activate carbon–halogen bonds in aryl halides, in much a similar way as that of its PCP analogue. Complex (3+) was also seen to establish a linear ↔ bent nitrosyl equilibrium upon addition of CO which could not be fully displaced with excess CO.


 Please wait while we load your content...
Please wait while we load your content...