Efficient alkane hydroxylation catalysis of nickel(ii) complexes with oxazoline donor containing tripodal tetradentate ligands†
Abstract
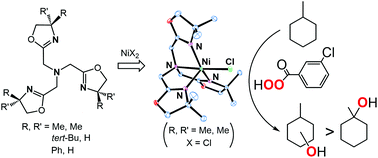
Tris(oxazolynylmethyl)amine TOAR (where R denotes the substituent groups on the fourth position of the oxazoline rings) complexes of nickel(II) have been synthesized as catalyst precursors for alkane oxidation with meta-chloroperoxybenzoic acid (m-CPBA). The molecular structures of acetato, nitrato, meta-chlorobenzoato and chlorido complexes with TOAMe2 have been determined using X-ray crystallography. The bulkiness of the substituent groups R affects the coordination environment of the nickel(II) centers, as has been demonstrated by comparison of the molecular structures of chlorido complexes with TOAMe2 and TOAtBu. The nickel(II)-acetato complex with TOAMe2 is an efficient catalyst precursor compared with the tris(pyridylmethyl)amine (TPA) analogue. Oxazolynyl donors’ strong σ-electron donating ability will enhance the catalytic activity. Catalytic reaction rates and substrate oxidizing position selectivity are controlled by the structural properties of the R of TOAR. Reaction of the acetato complex with TOAMe2 and m-CPBA yields the corresponding acylperoxido species, which can be detected using spectroscopy. Kinetic studies of the decay process of the acylperoxido species suggest that the acylperoxido species is a precursor of an active species for alkane oxidation.


 Please wait while we load your content...
Please wait while we load your content...