Accelerating hydrogen evolution in Ru-doped FeCoP nanoarrays with lattice distortion toward highly efficient overall water splitting†
Abstract
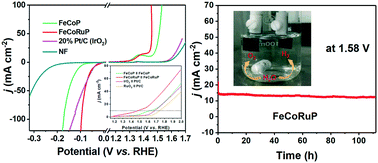
Rationally designing bifunctional electrocatalysts for the hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) with high activity and long term stability remains to be a big challenge. Herein, we report in situ synthesized uniform Ru-doped FeCoP (FeCoRuP) nanoarrays as a bifunctional electrocatalyst for highly efficient overall water splitting. The introduction of Ru modifies the electronic interaction between cation active sites due to the electronegativity difference, which also introduces more active sites by creating lattice defects and distortion. Benefiting from the rich active sites and the strong electronic interaction, the optimized Ru-doped FeCoP exhibits excellent HER activities (45 mV at 10 mA cm−2) and OER activity (214 mV at 20 mA cm−2), together with excellent long-term durability (110 h) in alkaline media. An overall water splitting cell in which both anode and cathode are composed of the FeCoRuP catalyst exhibits an ultralow voltage of 1.47 V at 10 mA cm−2, which is superior to the benchmark 20% Pt/C‖IrO2 electrodes (1.68 V at 10 mA cm−2). The present work offers a simple but effective approach to promote the overall water splitting performance of transition metal phosphides by modulating the electronic structure of active sites.


 Please wait while we load your content...
Please wait while we load your content...