Steam reforming of formaldehyde for generating hydrogen and coproducing carbon nanotubes for enhanced photosynthesis†
Abstract
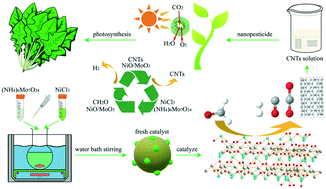
Nickel based catalysts exhibit high catalytic activity, but carbon deposition (coke poisoning) occurs in the field of steam reforming. To solve the coke poisoning of nickel based catalysts, a NiO/MoO3 catalyst was used for the formaldehyde steam reforming reaction, and coproduced carbon nanotubes (CNTs) were exploratively applied to improve plant photosynthesis. Results showed that the NiO/MoO3 catalyst exhibited the highest H2 selectivity of 104.2% at 600 °C, and its initiation temperature was 300 °C. Its formaldehyde conversion was higher than 90.0% at 400–600 °C. The synergistic effect between NiO and MoO3 guaranteed strong solid acidity, good redox properties and high concentration of surface oxygen species. It made NiO/MoO3 exhibit excellent hydrogen production performance. Furthermore, the in situ DRIFT study proved that the formaldehyde species adsorbed on the catalyst surface reacted with hydroxyl groups and water vapor also participated in the reaction. Then carbon monoxide (or carbon dioxide) and hydrogen formed by the decomposition of the intermediate. Finally, the crudely purified CNTs could be obtained by simple physical separation from the NiO/MoO3 catalyst and carbon deposits. The photosynthesis experiment also proved that the crudely purified CNTs could be used in the field of nanopesticides. They increased the plant yield by promoting plant photosynthesis.


 Please wait while we load your content...
Please wait while we load your content...