Domino lignin depolymerization and reconnection to complex molecules mediated by boryl radicals†
Abstract
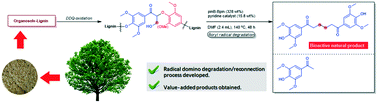
Chemical degradation of lignin has attracted increasing interest due to its potential for producing chemicals from renewable resources. Herein, we present a new transition metal free degradation procedure utilizing DDQ-oxidation and boryl radical mediated degradation, followed by reconnection of a monomer intermediate to a new dimer in a domino process. Our results include the selective degradation of oxidized β-O-4 model compounds by a boryl radical initiated with a catalytic amount of 4-(4-pyridinyl)benzonitrile and bispinacolborane B2(pin)2 as well as its application to organosolv lignin. This sequential procedure expands the toolbox for lignin degradation from simple depolymerization to high-value products by incorporating bond forming transformations within the process, and also provides a new transition-metal free method for the construction of 1,6-diketone fragments.


 Please wait while we load your content...
Please wait while we load your content...