Synthesis and molecular structure of model silica-supported tungsten oxide catalysts for oxidative coupling of methane (OCM)†
Abstract
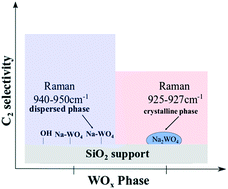
The molecular and electronic structures and chemical properties of the active sites on the surface of supported Na2WO4/SiO2 catalysts used for oxidative coupling of methane (OCM) are poorly understood. Model SiO2-supported, Na-promoted tungsten oxide catalysts (Na–WOx/SiO2) were systematically prepared using various Na- and W-precursors using carefully controlled Na/W molar ratios and examined with in situ Raman, UV-vis DR, CO2-TPD-DRIFT and NH3-TPD-DRIFT spectroscopies. The traditionally-prepared catalysts corresponding to 5% Na2WO4 nominal loading, with Na/W molar ratio of 2, were synthesized from the aqueous Na2WO4·2H2O precursor. After calcination at 800 °C, the initially amorphous SiO2 support crystallized to the cristobalite phase and the supported sodium tungstate phase consisted of both crystalline Na2WO4 nanoparticles (Na/W = 2) and dispersed phase Na–WO4 surface sites (Na/W < 2). On the other hand, the catalysts prepared via a modified impregnation method using individual precursors of NaOH + AMT, such that the Na/W molar ratio remained well below 2, resulted in: (i) SiO2 remaining amorphous (ii) only dispersed phase Na–WO4 surface sites. The dispersed Na–WO4 surface sites were isolated, more geometrically distorted, less basic in nature, and more reducible than the crystalline Na2WO4 nanoparticles. The CH4 + O2-TPSR results reveal that the isolated, dispersed phase Na–WO4surface sites were significantly more C2selective, but slightly less active than the traditionally-prepared catalysts that contain crystalline Na2WO4 nanoparticles (Na/W = 2). These findings demonstrate that the isolated, dispersed phase Na–WO4 sites on the SiO2 support surface are the selective-active sites for the OCM reaction.


 Please wait while we load your content...
Please wait while we load your content...