Bilateral photocatalytic mechanism of dye degradation by a designed ferrocene-functionalized cluster under natural sunlight†
Abstract
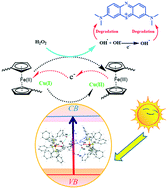
Extensive composition engineering research has been conducted on bandgap tunability, but the combination of two mechanisms for better photon harvesting over a wide range has rarely happened; this is of great importance for improving photocatalytic efficiency with sunlight. In order to enable concurrent heterogenic Fenton and Fenton-like reactions for dye degradation, two novel ferrocene-functionalized clusters, [(PPh3)3CuO2CFcCO2Cu(PPh3)3]·3CH3OH (D1) and [(PPh3)2AgO2CFcCO2Ag(PPh3)2]2·7CH3OH (D2) were designed, synthesized and characterized by multiple techniques. These chemically and thermally stable coinage clusters exhibit high photocatalytic activity towards the degradation of methylene blue as a model dye in the presence of H2O2 under direct sunlight irradiation. The degradation performance of complex D1 is about twice that of complex D2. The catalytic performance of D1 (15 000 mg g−1 in less than 20 min) is superior to those of other reported complexes, which can be attributed to the high level of generated hydroxyl radicals which are the most active species for dye degradation in the combination of Fenton and Fenton-like mechanisms. In addition to the degradation carried out with the aid of the Fe(III) of ferrocene, based on the Fenton mechanism, the photogenerated holes trapped by Cu(I) act as catalysts in the Fenton-like mechanism to produce an excess of hydroxyl radicals, adding to those formed via scavenging of photogenerated electrons by hydrogen peroxide. Furthermore, the performance of D1 in the presence of H2O2 as a dual photocatalyst under natural sunlight irradiation needs no pH adjustment which is a unique characteristic. This bilateral compound offers a promising strategy for the design of new photocatalysts.
- This article is part of the themed collection: Photocatalysis


 Please wait while we load your content...
Please wait while we load your content...