Heteroatom substituted zeolite FAU with ultralow Al contents for liquid-phase oxidation catalysis†
Abstract
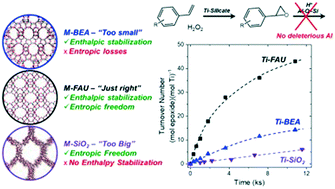
Heteroatom framework-substituted zeolites are important materials that enable shape- and size-selective catalysis. The efficacy of these materials for desired catalytic reactions depends critically on dispersive interactions between the microporous void of the zeolite and the reactant molecules stabilized within it. Here, we develop a post-synthetic method to synthesize base and transition metal-substituted (Ti, Nb, Ta, and Sn) FAU with ultralow Al contents (Si : Al > 900), which is confirmed using X-ray diffraction, elemental analysis, and N2 volumetric adsorption and 29Si MAS-NMR, DRUV-vis, and IR spectroscopic characterization. Turnover rates for styrene (C8H8) epoxidation within Ti-FAU are 2- and 7-fold greater than in analogous Ti-BEA and Ti–SiO2, respectively; yet, turnover rates of H2O2 decomposition are similar for all three materials. Consequently, Ti-FAU gives greater rates and selectivities for this reaction than common Ti-bearing silicates. The mechanism for epoxidation remains constant for all Ti-silicates examined (i.e., Ti-FAU, Ti-BEA, and Ti–SiO2). Therefore, the improved performance of Ti-FAU reflects differences in activation free energies for epoxidation that show an enthalpic preference in Ti-FAU relative to Ti–SiO2 and an entropic gain relative to Ti-BEA. These results demonstrate the synthesis of M-FAU with ultralow Al contents are useful for catalytic reactions involving bulky reactants that can not occur in smaller pore zeotype materials (Ti-MFI), that exhibit deactivation due to changes in Ti-atom coordination (e.g., Ti–SiO2), and that are prone to losses catalyzed by residual Brønsted acid sites (e.g., epoxidations, oxidations, and isomerization reactions).
- This article is part of the themed collection: 2020 Catalysis Science & Technology Hot Articles


 Please wait while we load your content...
Please wait while we load your content...