Electron-donating strength dependent symmetry breaking charge transfer dynamics of quadrupolar molecules†
Abstract
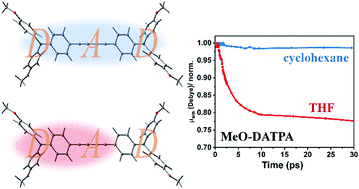
The excited state symmetry breaking charge transfer (SBCT) dynamics of two diacetylide-triphenylamine (DATPA) derivatives with different electron-donating abilities are investigated by femtosecond transient absorption and fluorescence spectroscopy. By tracking the evolution of the excited states by transient absorption spectra and the kinetics of the instantaneous emission dipole moments obtained from transient fluorescence spectroscopy, it is found that, in nonpolar solvent, the relaxed S1 state is quadrupolar with little change of emission dipole moments for the two molecules within 30 ps, whereas in polar solvent, the quadrupolar state evolves to a symmetry broken S1 state, in which, the emission dipole moment exhibits a fast reduction in the first few picoseconds. The larger reduction in emission transition dipole moment for the molecule with stronger electron-donating methoxy groups indicates a larger extent of symmetry breaking compared with the one with weak electron-donating methyl groups. Consequently, we revealed that the magnitude of symmetry breaking can be tuned by changing the electron-donors in quadrupolar molecules.


 Please wait while we load your content...
Please wait while we load your content...