C–H⋯S interaction exhibits all the characteristics of conventional hydrogen bonds†
Abstract
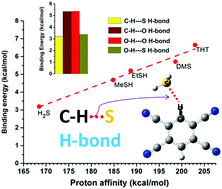
This is a tale of a pair of a hydrogen bond donor and acceptor, namely the CH donor and sulphur acceptor, neither of which is a conventional hydrogen bond participant. Sulfur (S), being less electronegative (2.58) compared to its first row analogue oxygen (3.44), has not been considered as a potential HB acceptor for a long time. The C–H⋯Y (Y = HB acceptor) interaction has its own history of exhibiting omnidirectional shifts in the CH stretching frequency upon complex formation. Therefore, a systematic investigation of the C–H⋯S interaction was the primary goal of the work presented here. Together with gas-phase vibrational spectroscopy and ab initio quantum chemical calculations, the nature and strength of the C–H⋯S hydrogen bond (HB) have been investigated in the complexes of 1,2,4,5-tetracyanobenzene (TCNB) with various sulfur containing solvents. Despite the unconventional nature of both HB donor and HB acceptor (C–H and S, respectively), it was found that the C–H⋯S hydrogen bond exhibits all the characteristics of the conventional hydrogen bond. The binding strength of the C–H⋯S H-bond in these complexes was found to be comparable to that of the conventional hydrogen bonds. The unusual stabilities of these HBs have been mainly attributed to the attractive dispersion interaction.


 Please wait while we load your content...
Please wait while we load your content...