Adsorption and oxidation of SO2 on the surface of TiO2 nanoparticles: the role of terminal hydroxyl and oxygen vacancy–Ti3+ states†
Abstract
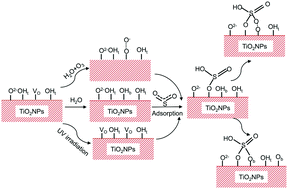
Herein, the absorption and oxidation reactions of SO2 on TiO2 nanoparticles (TiO2 NPs) at 296 K under various environmental conditions (humidity, UV irradiation, and ozone copresence) were investigated by using a flow chamber reaction system, synchrotron X-ray absorption near-edge structure (XANES) and high resolution synchrotron X-ray photoelectron spectroscopy (XPS) measurements. The results showed that oxidation of SO2 to sulfate via TiO2 NP catalysis happened at a very rapid rate. The appropriate relative humidity, UV irradiation and co-presence of ozone all markedly promoted SO2 oxidation on TiO2 NPs. High resolution XPS unraveled that the terminal hydroxyl (OHt) and oxygen vacancy (VO)–Ti3+ states on TiO2 NPs were the active sites for SO2 adsorption and oxidation. The data of XPS measurements suggest that SO2 was adsorbed on a OHt next to a Ti3+ VO and reacted to form HSO3−. HSO3− can then transform into SO32−via transfer of a proton. The resulting adsorbed SO32− could bind to a surface bridging O (Ob) atom and transform into SO42−. A H2O molecule could dissociate on VO–Ti3+ into two bridging hydroxyl (OHb) groups, subsequently forming new Ob, which provides an active O site for the adsorbed HSO3−/SO32− and oxidizes them into HSO4−/SO42− on the surface of the TiO2 NPs. The copresence of O3 could promote H2O dissociation into OHb, promoting the formation of Ob. The copresence of O3 may also promote the dissociation of adsorbed H2O into TiO2–O2− and hydroxyl radicals (˙OH) on VOs, facilitating the oxidation of adsorbed HSO3−/SO32−. Under UV irradiation, new VOs were created via oxidation of lattice O by photo-generated holes, resulting in increased Ob and subsequently enhanced oxidation of adsorbed HSO3−/SO32− on TiO2 NPs.


 Please wait while we load your content...
Please wait while we load your content...