Effect of the substituent position on the electrochemical, optical and structural properties of donor–acceptor type acridone derivatives†
Abstract
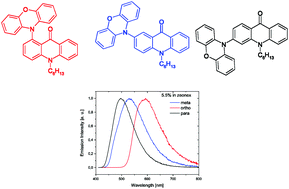
Three new donor–acceptor (D–A) compounds, positional isomers of phenoxazine-substituted acridone, namely 1-phenoxazine-N-hexylacridone (o-A), 2-phenoxazine-N-hexylacridone (m-A) and 3-phenoxazine-N-hexylacridone (p-A), were synthesized. The synthesized compounds showed interesting, isomerism-dependent electrochemistry. Their oxidation was reversible and their potential (given vs. Fc/Fc+) changed from 0.21 V for o-A to 0.36 V for p-A. In contrast, their reduction was irreversible, isomerism-independent and occurred at rather low potentials (ca. −2.25 to −2.28 V). The electrochemical results led to the following values of the ionization potentials (IPs) and electron affinities (EAs): 5.03 eV and −2.14 eV, 5.15 eV and −2.20 eV, and 5.20 eV and −2.28 eV for o-A, m-A and p-A, respectively. The experimentally obtained values were in very good agreement with those predicted by DFT calculations. All three isomers readily formed single crystals suitable for their structure determination. o-A and p-A crystallized in P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) and P21/n space groups, respectively, with one molecule per asymmetric unit, while m-A crystallized in the P21/c space group with two molecules in the asymmetric unit accompanied by disordered solvent molecules. The UV-vis spectra of the studied compounds were isomerism and solvent independent, yielding absorption maxima in the vicinity of 400 nm. Their photoluminescence spectra, in turn, strongly depended on isomerism and the used solvent showing smaller Stokes shifts for the emission bands registered in toluene as compared to the corresponding bands measured in dichloromethane. The photoluminescence quantum yields (ϕ) were systematically higher for toluene solutions reaching the highest value of 20% for p-A. For all three isomers studied, stationary and time-resolved spectroscopic investigations carried out in toluene at different temperatures revealed spectral features indicating a contribution of thermally activated delayed fluorescence (TADF) to the observed spectroscopic behaviour. The measured photoluminescence quantum yields (ϕ) were higher for solid state films of pure compounds and for their dispersions in solid matrices (zeonex) than those recorded for toluene and dichloromethane solutions of the studied phenoxazine-N-hexylacridone isomers. The obtained experimental spectroscopic and structural data were confronted with theoretical predictions based on DFT calculations.
and P21/n space groups, respectively, with one molecule per asymmetric unit, while m-A crystallized in the P21/c space group with two molecules in the asymmetric unit accompanied by disordered solvent molecules. The UV-vis spectra of the studied compounds were isomerism and solvent independent, yielding absorption maxima in the vicinity of 400 nm. Their photoluminescence spectra, in turn, strongly depended on isomerism and the used solvent showing smaller Stokes shifts for the emission bands registered in toluene as compared to the corresponding bands measured in dichloromethane. The photoluminescence quantum yields (ϕ) were systematically higher for toluene solutions reaching the highest value of 20% for p-A. For all three isomers studied, stationary and time-resolved spectroscopic investigations carried out in toluene at different temperatures revealed spectral features indicating a contribution of thermally activated delayed fluorescence (TADF) to the observed spectroscopic behaviour. The measured photoluminescence quantum yields (ϕ) were higher for solid state films of pure compounds and for their dispersions in solid matrices (zeonex) than those recorded for toluene and dichloromethane solutions of the studied phenoxazine-N-hexylacridone isomers. The obtained experimental spectroscopic and structural data were confronted with theoretical predictions based on DFT calculations.
- This article is part of the themed collection: 2020 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...