The role of non-covalent intermolecular interactions on the diversity of crystal packing in supramolecular dihalopyridine–silver(i) nitrate complexes†‡
Abstract
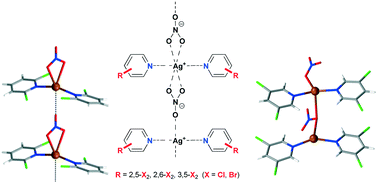
Six novel 1 : 2 complexes of silver(I) nitrate with 2,5-, 2,6- and 3,5-dihalopyridines (halo stands for chlorine and bromine) were prepared by the reaction of an aqueous solution of AgNO3 and the corresponding pyridine derivative in a water–alcohol solution. In spite of having analogous empirical formulae, they reveal intriguing structural differences, except [Ag(NO3)(2,5-Cl2py)2]n (1) and [Ag(NO3)(2,5-Br2py)2]n (4), which are isostructural. In contrast, [Ag(NO3)(3,5-Cl2py)2]2 (3) is dimeric, while its Br analogue [Ag(NO3)(3,5-Br2py)2]n (6) is polymeric in nature, being the only complex with no halogen⋯halogen interactions. Lastly, both [Ag(NO3)(2,6-Cl2py)2] (2) and [Ag(NO3)(2,6-Br2py)2] (5) consist of monomeric molecules, with dissimilar interaction patterns among units linking chains into different 3D structures. In addition, all isolated complexes were identified by elemental analysis and characterized in solution by multinuclear NMR spectroscopy (1H, 13C, 15N) and IR and mass spectrometry. DFT calculations verified the predominant tendency of the Ag+ ions to bind two pyridine nitrogen atoms and one nitrate ion in their complexes and confirm that their supramolecular structure is a compromise between competing weak intramolecular interactions and steric constrains.


 Please wait while we load your content...
Please wait while we load your content...