O–I–O halogen bond of halonium ions†
Abstract
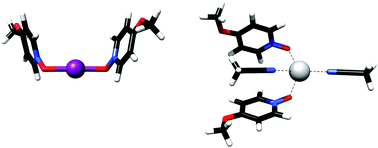
The reactivity of halonium ions is conveniently modulated by three-center, four-electron halogen bonds. Such stabilized halonium complexes are valuable reagents for oxidations and halofunctionalization reactions. We report the first example of the stabilization of a halenium ion in a three-center, four-electron halogen bond with two oxygen ligands. The influence of electron density and solvent on the stability of the complexes is assessed. O–I–O halogen bond complexes are applicable as synthetic reagents and as supramolecular synthons.


 Please wait while we load your content...
Please wait while we load your content...