Lateral flow fluorescent immunoassay based on isothermal amplification for rapid quantitative detection of Salmonella spp.
Abstract
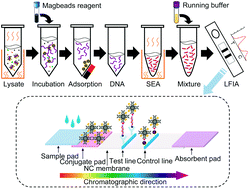
Salmonella spp. are zoonotic pathogens of substantial public health concern. To enable detection in the field or under instrument-free conditions, we developed a rapid and robust lateral flow fluorescent immunoassay based on strand exchange amplification (SEA-LFIA) for the quantitative detection of Salmonella spp. As far as we know, this work is the first report regarding the use of Bst DNA polymerase-assisted SEA for fluorescence sensing to detect Salmonella spp. The SEA method was further confirmed by enzymatic digestion and Sanger dideoxy sequencing. The specificity of SEA-LFIA assay was verified by 89 Salmonella strains (18 Salmonella reference strains and 71 clinical isolates) and 15 non-Salmonella reference strains (different genera). The sensitivity of SEA-LFIA assay was 6 × 100 CFU mL−1 of Salmonella pure culture or 3 × 104 CFU 25 g−1 of artificially spiked raw chicken meat. Using this assay, it was found that 37 (16%) of the 236 samples collected were positive, which was consistent with the results of conventional PCR. The cutoff value is 15 and SEA-LFIA assay only takes ∼30 min without high equipment and reagent cost. In addition, the proposed strategy can be easily extended by redesigning the corresponding amplification primers to detect target analytes. In conclusion, the optimized SEA-LFIA assay is an efficient and specific method for the detection of Salmonella spp., and can potentially serve as a new on-site diagnostic tool in life sciences.


 Please wait while we load your content...
Please wait while we load your content...