The effect of cation size on hydride-ion conduction in LnSrLiH2O2 (Ln = La, Pr, Nd, Sm, Gd) oxyhydrides†
Abstract
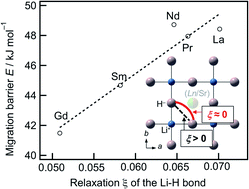
Hydride-ion conductors have attracted great interest as solid electrolytes due to the highly negative redox potential of the hydride ion. This study provides direction for effective design of materials that enhance H− conductivity by exploring the relationship between cation size and hydride-ion conductivity using LnSrLiH2O2 (Ln = La, Pr, Nd, Sm, Gd), which are K2NiF4-type oxyhydride materials, synthesised under ambient and high pressures. The size of the A-site cation and the activation energy for H− conduction were found to be strongly correlated. GdSrLiH2O2 exhibits the lowest activation energy of 67 kJ mol−1. Crystal structure analysis and first principles calculations revealed that H− is more displaced along the conduction pathway for smaller A-site cations in LnSrLiH2O2 due to lower repulsion between A-site cations and H−. These findings contribute to the further development of K2NiF4-type oxyhydrides with high H− conductivities.


 Please wait while we load your content...
Please wait while we load your content...