Arrangement of water molecules and high proton conductivity of tunnel structure phosphates, KMg1−xH2x(PO3)3·yH2O†
Abstract
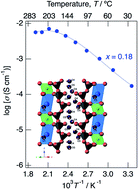
A fast proton conductor was investigated in a mixed-valence system of phosphates with a combination of large cations (K+) and small cations (Mg2+), which resulted in a new phase with a tunnel structure suitable for proton conduction. KMg1−xH2x(PO3)3·yH2O was synthesized by a coprecipitation method. A solid solution formed in the range of x = 0–0.18 in KMg1−xH2x(PO3)3·yH2O. The structure of the new proton conductor was determined using neutron and X-ray diffraction measurements. KMg1−xH2x(PO3)3·yH2O has a tunnel framework composed of face-shared (KO6) and (MgO6) chains, and PO4 tetrahedral chains along the c-direction by corner-sharing. Two oxygen sites of water molecules were detected in the one-dimensional tunnel, one of which exists as a coordination water of K+ sites. Multi-step dehydration was observed at 30 °C and 150 °C from thermogravimetric/differential thermal analysis measurements, which reflects the different coordination environments of the water of crystallization. Water molecules are connected to PO4 tetrahedra by hydrogen bonds and form a chain along the c-axis in the tunnel, which would provide an environment for fast proton conduction associated with water molecules. The KMg1−xH2x(PO3)3·yH2O sample with x = 0.18 exhibited high proton conductivity of 4.5 × 10−3 S cm−1 at 150 °C and 7.0 × 10−3 S cm−1 at 200 °C in a dry Ar gas flow and maintained the total conductivity above 10−3 S cm−1 for 60 h at 150 °C under N2 gas atmosphere.


 Please wait while we load your content...
Please wait while we load your content...