Photo-production of reactive oxygen species and degradation of dissolved organic matter by hematite nanoplates functionalized by adsorbed oxalate†
Abstract
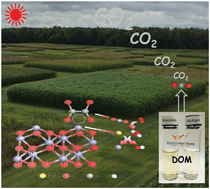
The geochemical cycling of iron and carbon can couple in unique ways in the euphotic zone of aquatic systems. For example, the prevalence of hematite nanoparticles and low molecular weight organics that can functionalize their surfaces, such as oxalate, creates a solar-photoactive interfacial system capable of generating reactive oxygen species (ROS) that degrade natural organic matter. Here we report a systematic study of this pathway and its efficiency to mineralize model chromophoric dissolved organic matter (DOM) compounds into CO2 under mildly acidic conditions. When illuminated, synthetic hematite nanoplates coated with adsorbed oxalate undergo ligand-to-metal charge transfer yielding Fe(II) via photoreductive dissolution, while oxalate decomposes into the carboxyl anion radical as detected by electron paramagnetic spectroscopy using selective spin trap compounds. This important radical quickly reduces molecular oxygen into hydrogen peroxide, which initiates the canonical Fenton reactions that yield the hydroxyl radical via Fe(II)/Fe(III) valence cycling. Production of hydroxyl radical in this photocatalytic system is shown to efficiently degrade the pseudo-DOM chromophore RhB, as well as two natural water-extractable chromophoric DOM compounds from agricultural soils, characterized in molecular detail using Fourier transform ion cyclotron resonance mass spectrometry, infrared fluorescence and nuclear magnetic resonance spectroscopies. The study highlights the photocatalytic interactions that can occur between common iron oxide nanoparticles, low molecular weight dicarboxylic acids, and dissolved organic matter that can couple their steady-state fluxes in the euphotic zone, pointing to the importance of light-induced ROS generation as a key mechanism.


 Please wait while we load your content...
Please wait while we load your content...
