An optimised Cu(0)-RDRP approach for the synthesis of lipidated oligomeric vinyl azlactone: toward a versatile antimicrobial materials screening platform†
Abstract
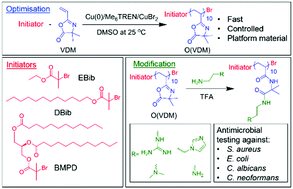
This report details the synthesis of lipidated 2-vinyl-4,4-dimethyl-5-oxazolone (VDM) oligomers via an optimised Cu(0)-mediated reversible-deactivation radical polymerisation approach, and the use of these oligomers as a versatile functional platform for the rapid generation of antimicrobial materials. The relative amounts of CuBr2 and Me6TREN were optimised to allow the fast and controlled polymerisation of VDM. These conditions were then used with the initiators ethyl 2-bromoisobutyrate, dodecyl 2-bromoisobutyrate, and (R)-3-((2-bromo-2-methylpropanoyl)oxy)propane-1,2-diyl didodecanoate to synthesise a library of oligo(VDM) (degree of polymerisation = 10) with ethyl, dodecyl or diglyceride end-groups. Subsequently, ring-opening of the pendant oxazolone group with various amines (i.e., 2-(2-aminoethyl)-1,3-di-Boc-guanidine, 1-(3-aminopropyl)imidazole, N-Boc-ethylenediamine, or N,N-dimethylethylenediamine) expanded the library to give 12 functional oligomers incorporating different cationic and lipid elements. The antimicrobial activities of these oligomers were assessed against a palette of bacteria and fungi: i.e. Staphylococcus aureus, Escherichia coli, Candida albicans, and Cryptococcus neoformans. The oligomers generally exhibited the greatest activity against the fungus, C. neoformans, with a minimum inhibitory concentration of 1 μg mL−1 (comparable to the clinically approved antifungal fluconazole). To assess haemocompatibility, the oligomers were assayed against erythrocytes, with the primary amine or guanidine containing C12 and 2C12 oligomers exhibiting greater lysis against the red blood cells (HC10 values between 7.1 and 43 μg mL−1) than their imidazole and tertiary amine counterparts (HC10 of >217 μg mL−1). Oligomers showed the greatest selectivity for C. neoformans, with the C12- and 2C12-tertiary amine and C12-imidazole oligomers possessing the greatest selectivity of >54–109. These results demonstrate the utility of reactive oligomers for rapidly assessing structure–property relationships for antibacterial and antifungal materials.


 Please wait while we load your content...
Please wait while we load your content...