Tetraphenylethene decorated with disulfide-functionalized hyperbranched poly(amido amine)s as metal/organic solvent-free turn-on AIE probes for biothiol determination†
Abstract
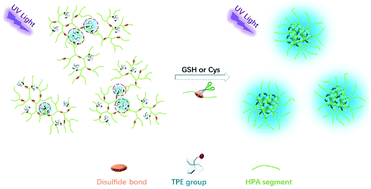
The development of “turn-on” fluorescence probes for biological thiol detection has received great attention in recent years because abnormal levels of these mercapto biomolecules are always related to some form of disease. In this work, a series of AIE-active polymer probes for biothiol detection were designed and synthesized, comprising disulfide-functionalized hyperbranched poly(amido amine) (ssHPA) as the hydrophilic matrix, tetraphenylethene (TPE) as the hydrophobic AIE-active fluorogen and a disulfide bond as the cleavable functional linkage, defined as TPE-ssHPAs. When these polymers were targeted as “turn on” fluorescence probes for the detection of biothiols, no organic solvents or heavy metal ions were needed as the quencher to eliminate background signals because of the relatively rigid dendritic structure. Upon introducing glutathione or cysteine to an aqueous solution of TPE-ssHPAs, bright fluorescence occurred because of the collapsing of the dendritic structure, leading to the aggregation of TPE units. Further in vitro experiments verified that TPE-ssHPAs is a qualified probe for biothiol detection in living cells. In addition, this strategy of embedding AIEgens inside a relatively rigid hyperbranched polymer offers an effective solution to “pre-quench” the fluorescence of “turn-on” type AIE-active sensors.


 Please wait while we load your content...
Please wait while we load your content...