Vacancy engineering of AuCu cocatalysts for improving the photocatalytic conversion of CO2 to CH4†
Abstract
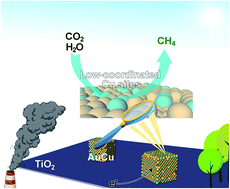
Photocatalytic conversion of CO2 and H2O to specific renewable fuels represents an appealing approach to abating the greenhouse effect and remedy the energy crisis. However, it often suffers from low yield and selectivity owing to charge recombination, poor reactivity as well as the occurrence of side reactions. Rational surface design of cocatalysts holds the key to address this grand challenge. In this article, the introduction of Cu vacancies onto the surface of cubic AuCu alloy cocatalysts is demonstrated to boost both the activity and selectivity in the photocatalytic conversion of CO2 with H2O to CH4. According to the experimental results, two effects contribute to the enhancement of CO2-to-CH4 conversion: (1) the removal of Cu improves the electron trapping ability of AuCu cocatalysts, which is instrumental to the 8-electron process of CH4 production; (2) low-coordinated surficial Cu sites neighboring the vacancies serve as highly selective sites for the reduction of CO2 to CH4. As a result, a highest selectivity of 94.7% for CH4 production with a maximal rate of 33.5 μmol gcat−1 h−1 is achieved by the vacancy-rich AuCu cocatalyst integrated with TiO2 nanosheets. This work provides fresh insights into engineering the surficial lattice of a cocatalyst at atomic precision for highly selective CO2 photoreduction through rational defect design.


 Please wait while we load your content...
Please wait while we load your content...