Green radicals of potassium poly(heptazine imide) using light and benzylamine†
Abstract
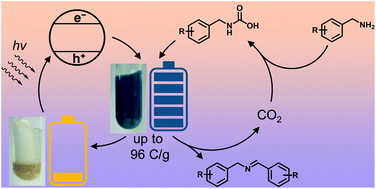
Tinted long-lived ionic carbon nitride radicals were recently introduced and applied in photocatalysis and energy storage. However, the reason for their higher activity in the photocatalytic reaction and optimal conditions for generating such radicals remain vague. Herein, we study the conditions for carbon nitride photocharging to achieve a higher charge density and validate a convenient method to quantify the number of electrons accumulated in carbon nitride semiconductors by quenching its radicals with methylviologen in the dark. In the presence of CO2, potassium poly(heptazine imide) (K-PHI) can be charged by up to 1000 μmol of electrons per gram of the material using benzylamine as an electron donor. Under the same conditions, mesoporous graphitic carbon nitride can accumulate only 50 μmol of electrons per gram. The products of the benzylamine oxidative coupling are imine and ammonia.


 Please wait while we load your content...
Please wait while we load your content...