Chemical-enzymatic fractionation to unlock the potential of biomass-derived carbon materials for sodium ion batteries†
Abstract
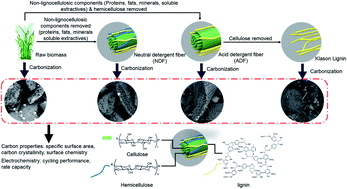
Plant biomass, the most abundant and sustainable carbon source, offers a rich chemical space to design hard carbons for sodium ion batteries. However, the compositional complexity of biomass has for a long time compromised the predictability of the structural and electrochemical properties of carbon. Using the chemical-enzymatic fractionation technique, we successively remove non-lignocellulosic components, hemicellulose and cellulose to create a suite of precursors for carbonization, in order to understand the roles of each biomass component in battery performance. Brewer's spent grain, an agricultural waste, is used as a representative biomass platform. The resulting hard carbon, with non-lignocellulosic components removed prior to carbonization, exhibits a dramatically reduced surface area and an increased specific capacity. Simultaneously removing non-lignocellulosic components and hemicellulose results in more sp2 carbon, expanded (002) interlayer spacing, and a remarkably improved specific capacity by four fold. Further removing cellulose, with only lignin remaining, significantly reduces the sp2 carbon and undermines the cycling stability of the derived carbon. Our finding reveals that the electrochemical properties of the biomass-derived hard carbons in sodium ion batteries may be positively correlated with cellulose and lignin but negatively impacted by non-lignocellulosic components and hemicellulose. Guided by this knowledge, we further fractionated two additional biomasses, i.e. grape pomace and walnut shells, for improving the carbon performance. After removing non-lignocellulosic components and hemicellulose, the resulting hard carbon delivers a reversible capacity of 296 mA h g−1 at 50 mA g−1 and retains 86.4% capacity after 200 cycles. Therefore, our results lay the foundation for unlocking the potential of biomass-derived carbon materials by precise fractionation of biomass components.


 Please wait while we load your content...
Please wait while we load your content...