A high-energy potassium–sulfur battery enabled by facile and effective imidazole-solvated copper catalysts†
Abstract
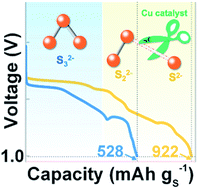
The potassium–sulfur (K–S) battery is a promising low-cost energy storage technology; however, its development is limited by a low achievable capacity (∼560 mAh gS−1) and a low average cell voltage (typically <1.5 V) owing to the formation of thermodynamic traps (K2S3) and sluggish reduction kinetics. In this study, we design a facile and effective homogeneous catalyst, 1-methylimidazole (Me-Im) solvated-copper, to promote the reduction kinetics of K2S3 to K2S, achieving a high reversible sulfur capacity of 922 mAh gS−1 with a high average cell voltage of 1.93 V, corresponding to a gravimetric energy of 1779 Wh kgS−1. X-ray photoelectron spectroscopy, X-ray diffraction and density functional theory calculation elucidated the working mechanism of the Me-Im-solvated Cu catalyst toward weakening the S–S bond in K2S3 and promoting the formation of K2S. This work provides a key strategy to achieve high-energy reversible potassium–sulfur batteries.


 Please wait while we load your content...
Please wait while we load your content...