Thermal degradation of formamidinium based lead halide perovskites into sym-triazine and hydrogen cyanide observed by coupled thermogravimetry-mass spectrometry analysis†
Abstract
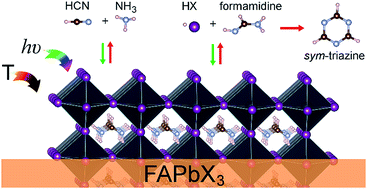
The thermal stability and decomposition products of formamidinium, a widely used organic cation in perovskite solar cell formulation, were investigated. The thermal degradation experiments of formamidinium-based perovskites and their halide precursors were carried out under helium atmosphere and vacuum at a constant heating rate of 20 °C min−1. In addition, pulsed heating steps were employed under illumination/dark conditions to simulate a more realistic working temperature condition for photovoltaic devices. The identification of gas decomposition products was based on the quadrupole mass spectrometry technique. The released amounts of sym-triazine, formamidine, and hydrogen cyanide (HCN) were observed to highly depend on the temperature. For the experimental conditions used in this study, sym-triazine was obtained as the thermal product of degradation at temperatures above 95 °C. Below this temperature, only formamidine and HCN generation routes were observed. The energy pathways of formamidinium thermal degradation under photovoltaic working temperature conditions were further assessed by density functional theory calculations. The results indicated that formamidinium was more resilient to thermal degradation and the release of irreversible decomposition products compared to methylammonium because of a larger enthalpy and activation energy obtained for the decomposition reactions. The HCN instantaneous concentration observed during the low temperature heating tests and the estimations of the maximum release of HCN achievable per meter-square of an FA based perovskite based solar cell were compared to acute exposure guideline levels of airborne HCN concentration.


 Please wait while we load your content...
Please wait while we load your content...