g-C3N4 templated synthesis of the Fe3C@NSC electrocatalyst enriched with Fe–Nx active sites for efficient oxygen reduction reaction†
Abstract
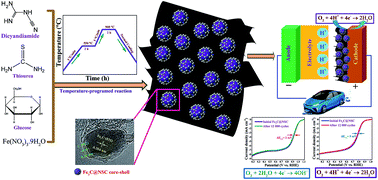
A porous, efficient, and durable nonprecious electrocatalyst for the oxygen reduction reaction (ORR) under both alkaline and acidic conditions is synthesized by a green, solvent-free, one-pot, economical, scalable, and template-free strategy. In situ formed graphitic carbon nitride (g-C3N4) is utilized as a sacrificial template for constructing the core–shell Fe3C@NSC electrocatalyst with a highly porous substructure and nanosheet-like morphology. The presence of glucose facilitates the homogeneous distribution of nanoparticles and establishes the nanosheet structure, otherwise agglomeration and formation of nanotubes occur in the Fe3C@NSC electrocatalyst. Optimizing the synthetic conditions generates a highly active ORR electrocatalyst with a unique mesoporous architecture, large surface area, high nitrogen doping level, encapsulated Fe3C nanoparticles in NS co-doped carbon layers and multiple exposed edge active centers. The best performing electrocatalyst displays outstanding ORR performance under alkaline conditions with onset potential and half-wave potential (E1/2) values of 1.059 and 0.938 V vs. the reversible hydrogen electrode (RHE), much higher than those of the state-of-the-art commercial Pt/C (20%), and admirable performance in acidic media. In addition, it exhibits 4e− selectivity, tolerance toward methanol, and superb durability with zero degradation after 12 000 potential cycles under both alkaline and acidic conditions. These exceptional characteristics make the as-synthesized electrocatalyst one of the top-level non-precious ORR electrocatalysts reported to date.


 Please wait while we load your content...
Please wait while we load your content...