Introducing pyridyl into electron transport materials plays a key role in improving electron mobility and interface properties for inverted perovskite solar cells†
Abstract
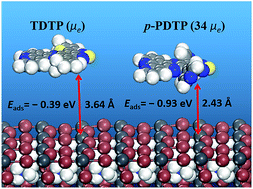
In inverted perovskite solar cells (PSCs), electron transfer materials (ETMs) have a significant effect on the efficiency and stability, because they can reduce the energy barrier between the perovskite film and electrode materials and suppress the potential interfacial charge recombination. Herein we designed a series of ETMs (o-PDTP, m-PDTP, and p-PDTP) through introducing pyridyl as the side chain of the experimentally synthesized ETM 10,14-bis(5-(2-ethylhexyl)thiophen-2-yl)-dipyrido-[3,2-a:2′,3′-c][1,2,5]-thiadiazolo-[3,4-i]-phenazine (TDTP) based on quantum chemical calculations. Our results showed that introducing pyridyl changes the molecular packing mode from the herringbone packing motif in TDTP to two-dimensional π–π stacking in p-PDTP due to the strong interactions between the added nitrogen atoms and the hydrogen atoms of the molecular skeleton. Therefore, the average electron mobility of p-PDTP (1.65 cm2 V−1 s−1) is 34 times higher than that of TDTP. Our results also indicated that the additional N⋯Pb interface interaction between the nitrogen atoms in the ETMs and the lead atom in MAPbI3 leads to stronger binding energies and easier electron injection. The present results not only offer two promising ETMs (m-PDTP and p-PDTP), but also propose a new design strategy of introducing pyridyl into ETMs for more efficient PSCs.


 Please wait while we load your content...
Please wait while we load your content...