Two-dimensional ZnO for the selective photoreduction of CO2†
Abstract
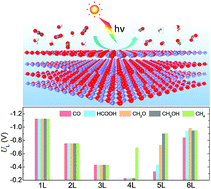
Ultrathin oxide films are technologically important for electronics, sensors and catalysis. When the film thickness is below some characteristic value, exotic electronic properties and catalytic behaviors would emerge. Understanding the thickness dependency of activity is crucial for controlling the catalytic performance of oxide films. Herein, we exploited two-dimensional (2D) zinc oxide (ZnO), whose freestanding sheets are available in the laboratory, as a prototype system for CO2 photoreduction. The evolution of structures and photocatalytic properties of ZnO sheets with the number of layers from one to eight is unveiled by first-principles calculations. Distinct from their bulk counterpart, the graphene-like non-polar structures of ZnO ultrathin films endow electron-accumulated surfaces. Not only do they have favorable band edge positions and optical absorbance, but the 2D ZnO sheets can also provide active sites for CO2 reduction with selectivity intrinsically depending on the number of layers and activity being correlated to the O 2p band center. The present results offer a universal strategy for tailoring the photocatalytic activity and selectivity of ultrathin films of metal oxides and other semiconductor compounds by adjusting the number of layers and taking advantage of the quantum confinement effect.


 Please wait while we load your content...
Please wait while we load your content...