Effect of eutectic accelerator in selenium-doped sulfurized polyacrylonitrile for high performance room temperature sodium–sulfur batteries†
Abstract
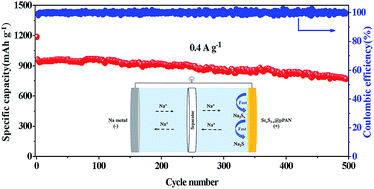
Sulfurized polyacrylonitrile is a suitable cathode candidate for room temperature sodium–sulfur batteries. However, its limited reactivity results in low utilization of active materials, limited rate capability and poor cycling performance especially in ether electrolyte. Here, we demonstrate the effect of using selenium as a eutectic accelerator in sulfurized polyacrylonitrile in which a small amount of selenium is easily distributed at the molecular level and leads to significant improvement of reaction kinetics. As a result, the designed Se0.08S0.92@pPAN cathode exhibits superior rate and cycle performance as well as compatibility with both ether and carbonate electrolytes, delivers capacities of 1214 and 767 mA h g−1 at 0.1 and 3 A g−1 respectively, and maintains a good specific capacity of 770 mA h g−1 at 0.4 A g−1 over 500 cycles (0.045% decay per cycle) in carbonate electrolyte with nearly 100% coulombic efficiency. Furthermore, self-discharge tests reveal diminished soluble sodium polysulfides owing to the fast redox conversion enabled by Se-doping.


 Please wait while we load your content...
Please wait while we load your content...