Host–guest interactions of catechol and 4-ethylcatechol with surface-immobilized blue-box molecules†
Abstract
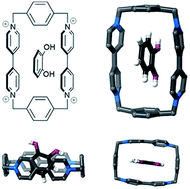
Catechol and 4-ethylcatechol are highly toxic, carcinogenic, and mutagenic molecules, often produced in water in small quantities as a result of hydrothermal upgrading processes. In this work, we demonstrate the facile immobilization of cyclobis(paraquat-p-phenylene) macrocycles (known as blue box (BB)) on silica surfaces, and show that BB-coated silica surfaces adsorb large amounts of catechol and 4-ethylcatechol, with a molar ratio of 214 : 1 and 432 : 1, respectively, instead of the expected 1 : 1 ratio. The adsorbates formed large aggregates on the silica surface, with a height range of 2–55 nm dependent on the catechol concentration and reaction time, while uncoated silica powder did not adsorb any catechol from water. The aggregates are shown to be poly(catechol)s, as a result of a BB-induced polymerization process in water under non-alkaline conditions. The process is initiated by the formation of radical cations of the adsorbed catechol and 4-ethylcatechol upon the complexation with assembled BB molecules through charge-transfer interactions. This is the first report of the polymerization of guest molecules catalyzed by surface-immobilized BB macrocycles. A BB-coated silica powder was used to extract up to 95% of 4-ethylcatechol in 3 hours and 60% of catechol in 2 hours from dilute aqueous solutions and did not leave particulates behind. More than 90% of the BB4+ macrocycles could be recovered from the silica surfaces after the water treatment processes.


 Please wait while we load your content...
Please wait while we load your content...