A robust solid oxide electrolyzer for highly efficient electrochemical reforming of methane and steam†
Abstract
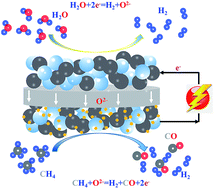
In this work, a robust solid oxide electrolysis cell with Sr2Fe1.5Mo0.5O6−δ–Ce0.8Sm0.2O1.9 (SFM–SDC) based electrodes has been utilized to verify the conceptual process of partial oxidation of methane (POM) assisted steam electrolysis, which can produce syngas and hydrogen simultaneously. When the cathode is fed with 74%H2–26%H2O and operated at 850 °C, the open circuit voltage (OCV), the minimum energy barrier required to overcome the oxygen partial gradient, is remarkably reduced from 0.940 to −0.012 V after changing the feed gas in the anode chamber from air to methane, indicating that the electricity consumption of the steam electrolysis process could be significantly reduced and compensated by the use of low grade thermal energy from external heat sources. It is found that after ruthenium (Ru) impregnation, the electrolysis current density of the electrolyzer is effectively enhanced from −0.54 to −1.06 A cm−2 at 0.6 V and 850 °C, while the electrode polarization resistance under OCV conditions and 850 °C is significantly decreased from 0.516 to 0.367 Ω cm2. Long-term durability testing demonstrates that no obvious degradation but a slight improvement is observed for the electrolyzer, which is possibly due to the activation of the SFM–SDC electrode during operation. These results indicate that the robust Ru infiltrated solid oxide electrolyzer is a very promising candidate for POM assisted steam electrolysis applications. Our result will provide insight to improve the electrode catalysts used in POM assisted steam electrolysis.


 Please wait while we load your content...
Please wait while we load your content...