Creating stable interfaces between reactive materials: titanium nitride protects photoabsorber–catalyst interface in water-splitting photocathodes†
Abstract
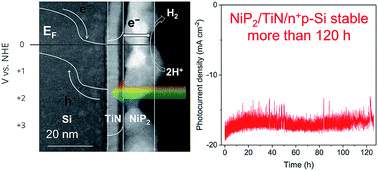
The development of a solar-driven water splitting device that replaces costly precious metals, while achieving stable high performance, is a major challenge. Transition metal phosphides are active and low-cost catalysts for the hydrogen evolution reaction (HER), although, none thus far have exhibited stable performance when interfaced with semiconductors. Here, we report on a monolithic junction consisting of cubic-NiP2:TiN:Si, fabricated using both commercial and custom Si photovoltaics. Stable performance is achieved using an ultrathin film of crystalline TiN that effectively hinders atomic diffusion between interfaces during fabrication. Crystalline cubic-NiP2 deposited on TiN/n+p-Si retains 97% of the bare Si photovoltage, comparable saturation current density to bare Si, and has a turnover frequency of 1.04 H2 per site per s at −100 mV applied potential. In acid, it requires only −150 mV additional overpotential compared to the benchmark, Pt/TiN/n+p-Si, to reach a HER photocurrent density of −10 mA cm−2. This photocathode maintains a stable H2 photocurrent (±10%) for at least 125 hours, the duration of testing. When the same layers are fabricated on a commercial Si solar cell, this photocathode produced double the photocurrent density (36.3 mA cm−2, under simulated 1.5 AM G illumination). Physical characterization gives detailed information on the properties responsible for the observed activity and durability of these interfaces. In general, the thin-film methodology presented here is widely applicable, demonstrates superior activity, and achieves long-term stability.


 Please wait while we load your content...
Please wait while we load your content...