Operation of proton exchange membrane (PEM) fuel cells using natural cellulose fiber membranes†
Abstract
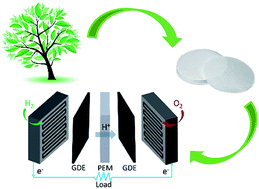
Proton exchange membranes (PEMs), such as Nafion, are still one of the reasons for the high cost of PEMFCs. Among the alternatives, cellulose, a low cost and biodegradable material, has been considered in the form of its derivatives, such as cellulose acetate, bacterial cellulose or nanocellulose, as the PEM in fuel cells. However, its use involves more chemical synthesis steps. Here, we use low-cost 1.5-micron cellulose filter paper as the scaffold and prepare membranes for PEMFCs by simply impregnating it with a 10% Nafion solution or immersing it in resorcinol bis(diphenyl phosphate) (RDP). The prepared membranes are primarily composed of cellulose, with a cellulose to Nafion or RDP ratio of 2 : 1 or 1 : 1. The membrane electrode assemblies (MEAs) incorporating the as-prepared membranes exhibit a maximum output power of 23 mW cm−2 at 80 °C for the cellulose/Nafion membrane or 10 mW cm−2 at 60 °C for the cellulose/RDP membrane with only 0.1 mg cm−2 Pt loading on the anode and cathode, when operated in air. The stability results of the membranes after 100 hours of continuous operation indicate a loss of only 10% in power for the cellulose/Nafion membrane and 20% for the cellulose/RDP membrane. It is revealed by FTIR spectroscopy that both the Nafion polymer and RDP were hydrogen bonded to the cellulose fibers, facilitating proton conduction and stabilizing them against further dissolution.
- This article is part of the themed collection: 2019 Sustainable Energy and Fuels HOT Articles


 Please wait while we load your content...
Please wait while we load your content...