Electrochemical C–H bond activation via cationic iridium hydride pincer complexes†
Abstract
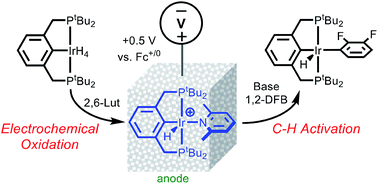
A C–H bond activation strategy based on electrochemical activation of a metal hydride is introduced. Electrochemical oxidation of (tBu4PCP)IrH4 (tBu4PCP is [1,3-(tBu2PCH2)-C6H3]−) in the presence of pyridine derivatives generates cationic Ir hydride complexes of the type [(tBu4PCP)IrH(L)]+ (where L = pyridine, 2,6-lutidine, or 2-phenylpyridine). Facile deprotonation of [(tBu4PCP)IrH(2,6-lutidine)]+ with the phosphazene base tert-butylimino-tris(pyrrolidino)phosphorane, tBuP1(pyrr), results in selective C–H activation of 1,2-difluorobenzene (1,2-DFB) solvent to generate (tBu4PCP)Ir(H)(2,3-C6F2H3). The overall electrochemical C–H activation reaction proceeds at room temperature without need for chemical activation by a sacrificial alkene hydrogen acceptor. This rare example of undirected electrochemical C–H activation holds promise for the development of future catalytic processes.
- This article is part of the themed collection: Celebrating our 2020 Prize and Award winners


 Please wait while we load your content...
Please wait while we load your content...