Diacetyl as a “traceless” visible light photosensitizer in metal-free cross-dehydrogenative coupling reactions†
Abstract
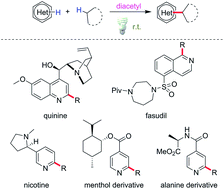
Minisci alkylation is of prime importance for its applicability in functionalizing diverse heteroarenes, which are core structures in many bioactive compounds. In alkyl radical generation processes, precious metal catalysts, high temperatures and excessive oxidants are generally involved, which lead to sustainability and safety concerns. Herein we report a new strategy using diacetyl (2,3-butanedione) as an abundant, visible light-sensitive and “traceless” hydrogen atom abstractor to achieve metal-free cross-dehydrogenative Minisci alkylation under mild conditions. Mechanistic studies supported hydrogen atom transfer (HAT) between an activated C(sp3)–H substrate and diacetyl. Moreover, with the assistance of di-tert-butyl peroxide (DTBP), the scope of the reaction could be extended to strong aliphatic C–H bonds via diacetyl-mediated energy transfer. The robustness of this strategy was demonstrated by functionalizing complex molecules such as quinine, fasudil, nicotine, menthol and alanine derivatives.
- This article is part of the themed collections: Most popular 2019-2020 organic chemistry articles and Most popular 2018-2019 organic chemistry articles


 Please wait while we load your content...
Please wait while we load your content...