Functionalization of polyacrylamide for nanotrapping positively charged biomolecules†
Abstract
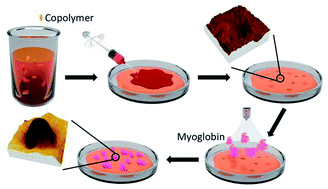
Engineering new materials which are capable of trapping biomolecules in nanoscale quantities, is crucial in order to achieve earlier diagnostics in different diseases. This article demonstrates that using free radical copolymerization, polyacrylamide can be successfully functionalized with specific synthons for nanotrapping positively charged molecules, such as numerous proteins, through electrostatic interactions due to their negative charge. Specifically, two functional random copolymers, acrylamide/acrylic acid (1) and acrylamide/acrylic acid/N-(pyridin-4-yl-methyl)acrylamide (2), whose negative net charges differ in their water solutions, were synthetized and their ability to trap positively charged proteins was studied using myoglobin as a proof-of-concept example. In aqueous solutions, copolymer 1, whose net charge for a 100 chain fragment (QpH 6/M) is −1.323 × 10−3, interacted with myoglobin forming a stable monodisperse nanosuspension. In contrast, copolymer 2, whose value of QpH 6/M equals −0.361 × 10−3, was not able to form stable particles with myoglobin. Nevertheless, thin films of both copolymers were grown using a dewetting process, which exhibited nanoscale cavities capable of trapping different amounts of myoglobin, as demonstrated by bimodal AFM imaging. The simple procedures used to build protein traps make this engineering approach promising for the development of new materials for biomedical applications where trapping biomolecules is required.


 Please wait while we load your content...
Please wait while we load your content...