Guest-mediated chirality transfer in the host–guest complexes of an atropisomeric perylene bisimide cyclophane host†‡
Abstract
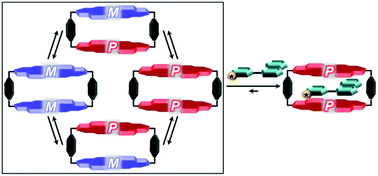
The dynamic equilibrium of stereoisomers of a cyclophane, which is comprised of two atropisomeric perylene bisimide (PBI) subunits, and its host–guest binding with an achiral and a series of chiral guests have been studied. Temperature-dependent 1H NMR spectroscopic studies have shown that at higher temperature all three stereoisomers ((M,M) and (P,P) enantiomers and (M,P)/(P,M) mesomers) of the cyclophane are in a dynamic equilibrium as only one set of proton signals was observed. However, at a lower temperature (260 K) two sets of signals in a ratio of 2 : 1, instead of the theoretically expected 1 : 1 ratio, were observed for the diastereomers (M,M)/(P,P) and (M,P)/(P,M) of the cyclophane presumably due to the chiral recognition of the enantiomeric (M,M)/(P,P) conformers. The 1H NMR studies further revealed that the achiral guest perylene mostly selects the (M,M)/(P,P) enantiomeric pair of the host for encapsulation. The induction of chirality in the host–guest complexes of the cyclophane with chiral guests has been investigated by circular dichroism (CD) spectroscopy. A distinct CD effect was observed upon addition of chiral guests to the solution of a racemic cyclophane host, indicating a pronounced preference of the chiral guests for one of the two homochiral stereoisomers, (M,M) and (P,P), for encapsulation. Kinetic studies by time-dependent NMR spectroscopy did not lead to conclusive results to assign the recognition process of chiral guests to either the induced fit or the conformational selection model.
- This article is part of the themed collection: In celebration of Julius Rebek’s 75th Birthday


 Please wait while we load your content...
Please wait while we load your content...