Cyanofluorination of vinyl ethers enabled by electron donor–acceptor complexes†
Abstract
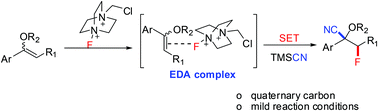
An efficient and mild three-component cyanofluorination of vinyl ethers with Selectfluor and TMSCN is described. The reaction protocol is operationally simple, allowing the access to various α-alkoxy-β-fluoronitriles bearing quaternary α-carbons in good to excellent yields. The mechanistic studies indicated that the formation of an electron donor–acceptor (EDA) aggregate and a subsequent single electron transfer (SET) process might be involved in the reaction.


 Please wait while we load your content...
Please wait while we load your content...