Bromobenzene aliphatic nucleophilic substitution guided controllable and reproducible synthesis of high quality cesium lead bromide perovskite nanocrystals†
Abstract
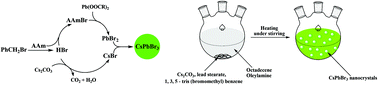
Because of the intrinsic low formation energies of CsPbBr3 perovskite nanocrystal (PNC) lattices, it is still a big challenge to controllably fabricate high quality CsPbBr3 PNCs with reproducibility. We develop a new chemical design for the controllable and reproducible synthesis of high quality CsPbBr3 PNCs based on bromobenzene and alkane amine aliphatic nucleophilic substitution (BANS). In this design, bromobenzene reacts with alkane amines under heating to release HBr and the HBr is trapped by the deacid reagent Cs2CO3 and alkane amines under heating to produce CsBr and alkane ammonium bromide (AAmBr). The intermediate product, AAmBr, reacted with lead stearate to generate PbBr2 that finally formed CsPbBr3 PNCs. The nucleation and growth kinetics of CsPbBr3 PNCs were easily controlled via regulating the reactivity of bromobenzene and alkane amine. Typically, when 1,3,5-tris(bromomethyl)benzene and oleylamine were used, CsPbBr3 PNCs with a quantum yield of 88% and a narrow full-width at half-maximum of 22 nm could be produced by loading all reactants into a single reaction vessel under continuous heating under ambient atmospheric conditions. The whole synthesis was completed within 10 min with high simplicity and reproducibility and could be amplified to gram scale synthesis without hindering the photoluminescence properties of CsPbBr3 PNCs. Moreover, the CsPbBr3 PNCs obtained have been successfully applied for wavelength shift based fluorescent colorimetric detection of hydrochloric acid with high selectivity and accuracy.


 Please wait while we load your content...
Please wait while we load your content...