Synthesis and self-assembly of photoacid-containing block copolymers based on 1-naphthol†
Abstract
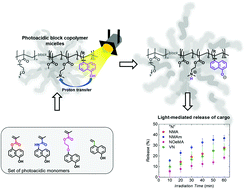
Photoacids experience a strong increase in acidity when absorbing light and, hence, can be considered as molecular switches. Herein, we present photo-responsive amphiphilic diblock quarterpolymers containing different photoacidic comonomers based on 1-naphthol (“N”). RAFT polymerization gives access to well-defined terpolymers containing methyl methacrylate (MMA/“M”), 2-(N,N-dimethylamino)ethyl methacrylate (DMAEMA/“D”), and a specific amount of the respective photoacidic comonomer. These materials are then used as macroinitiators for hydrophilic nona(ethylene glycol)methyl ether methacrylate (MEO9MA/“O”), followed by deprotection of the photoacidic comonomers to give the diblock quarterpolymer, P[(Mx-co-Dy-co-“N”z)-b-(Om)]. Afterwards, self-assembly in water as selective solvent leads to the formation of spherical diblock quarterpolymer micelles with controllable size. To demonstrate the potential of these photo-responsive micelles for controlled release, a model hydrophobic guest molecule (Nile Red) was encapsulated, and triggered release based on a light-mediated excited state proton transfer (ESPT) from the photoacid to DMAEMA could be observed, due to a shift in the hydrophilic/hydrophobic balance of the overall diblock quarterpolymer.


 Please wait while we load your content...
Please wait while we load your content...