Influence of different sequences of l-proline dipeptide derivatives in the pendants on the helix of poly(phenylacetylene)s and their enantioseparation properties†
Abstract
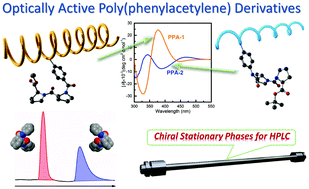
Two novel phenylacetylenes containing L-proline dipeptide derivatives with different sequences were synthesized and polymerized in THF using Rh(nbd)BPh4 as a catalyst to obtain the corresponding one-handed helical poly(phenylacetylene) derivatives (PPA-1 and PPA-2). The effect of different sequences of L-proline dipeptide derivatives in the pendants on the helical conformation of the polymer main chain was mainly evaluated and the enantioseparation properties of PPA-1 and PPA-2 for eleven racemates were investigated as chiral stationary phases (CSPs) for HPLC. In N,N-dimethylacetamide, the CD signals of PPA-1 and PPA-2 were the opposite, and the CD intensity of PPA-1 was higher than that of PPA-2, indicating that the two polymers possess different main chain helical conformations and PPA-1 may have a higher screw-sense excess. PPA-1 and PPA-2 showed rather different enantioseparation properties depending on the different helical structures, induced by different coating solvents and the different sequences of L-proline dipeptide derivatives in the pendants. In addition, each of the two poly(phenylacetylene) derivatives has its own merits. PPA-1 showed a more extensive chiral selectivity, while PPA-2 showed a more specific recognition of certain enantiomers, such as racemates 8 and 9. These two racemates could be better resolved on PPA-2 with the separation factors being even higher than those on the well-known commercial chiral columns Chiralcel or Chiralpak AD, OD, AS and OJ-H.


 Please wait while we load your content...
Please wait while we load your content...