Mechanism of fatty acid decarboxylation catalyzed by a non-heme iron oxidase (UndA): a QM/MM study†
Abstract
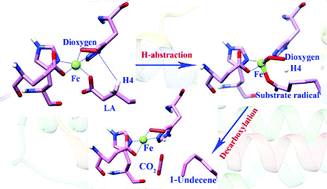
UndA is a non-heme iron enzyme that was recognized to catalyze the decarboxylation of medium chain (C10–C14) fatty acids to produce trace amounts of 1-alkenes. Owing to the electron imbalance during the oxidative decarboxylation of the substrate and the reduction of O2, only single turnover reactions were obtained in UndA in vitro assays. Unlike the general non-heme iron enzymes, the catalytic efficiency of UndA is quite low. According to the previous proposal, both FeIII–OO˙− and FeIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O complexes may abstract the β-H of fatty acids to trigger the oxidative decarboxylation reaction. Herein, on the basis of the crystal structures of UndA in complex with the substrate analogues, we constructed a series of computational models and performed quantum mechanics/molecular mechanics (QM/MM) calculations to explore the UndA-catalyzed decarboxylation using lauric acid as the substrate. Our calculation results reveal that only the FeIII–OO˙− complex can initiate the decarboxylation, and the substrate (lauric acid) should monodentately coordinate to the Fe center to facilitate the β-H abstraction. In addition, the monodentate coordination corresponds to higher relative energy than the bidentate mode, which may explain the low efficiency of UndA. It is also revealed that as long as the β-H is extracted by the FeIII–OO˙−, the decarboxylation of the substrate radical is quite easy, and an electron transfer from the substrate to the iron center is the prerequisite. For the FeIV
O complexes may abstract the β-H of fatty acids to trigger the oxidative decarboxylation reaction. Herein, on the basis of the crystal structures of UndA in complex with the substrate analogues, we constructed a series of computational models and performed quantum mechanics/molecular mechanics (QM/MM) calculations to explore the UndA-catalyzed decarboxylation using lauric acid as the substrate. Our calculation results reveal that only the FeIII–OO˙− complex can initiate the decarboxylation, and the substrate (lauric acid) should monodentately coordinate to the Fe center to facilitate the β-H abstraction. In addition, the monodentate coordination corresponds to higher relative energy than the bidentate mode, which may explain the low efficiency of UndA. It is also revealed that as long as the β-H is extracted by the FeIII–OO˙−, the decarboxylation of the substrate radical is quite easy, and an electron transfer from the substrate to the iron center is the prerequisite. For the FeIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O complex, since the β-H is far from the OFe atom and the angle of ∠Fe–O–H is 53.1°, the H-abstraction is calculated to be difficult.
O complex, since the β-H is far from the OFe atom and the angle of ∠Fe–O–H is 53.1°, the H-abstraction is calculated to be difficult.
- This article is part of the themed collection: Mechanistic, computational & physical organic chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...