Asymmetric total synthesis of cryptoconcatone I†
Abstract
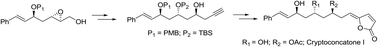
Asymmetric total synthesis of the naturally occurring cryptoconcatone I, which possesses a γ-Z-butenolide framework, is described here for the first time. Asymmetric propargylation, E-selective cross metathesis, and regioselective reductive epoxide ring opening were employed to access the enantiopure terminal alkyne. Late stage bimetallic cascade cyclization of the alkyne with (Z)-3-bromoacrylic acid afforded the natural product in an efficient way.
- This article is part of the themed collection: Total synthesis in OBC


 Please wait while we load your content...
Please wait while we load your content...