The mechanistic duality of (thio)urea organocatalysts for ring-opening polymerization
Abstract
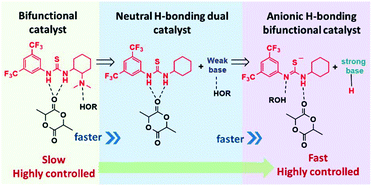
Among the various catalysts for ROP, H-bonding organocatalysts stand out in the precise level of reaction control they are able to render during ROP. The H-bonding class of organocatalysts are thought to effect ROP via dual activation of both monomer and chain end. (Thio)urea mediated ROP has experienced a renaissance as a new polymerization mechanism – mediated by imidate or thioimidate species – facilitates new modes of reactivity and new synthetic abilities. Indeed, the urea class of H-bond donors has been shown to be more active than their corresponding thioureas. The imidate mechanism remains highly active in polar solvents and exhibits remarkable control – and ‘living’ behavior – under solvent-free conditions, and a broad range of temperatures is accessible. The advancements in synthetic abilities have all evolved through a greater understanding of reaction mechanism. Through the continued synergistic advances of catalysis and material, the (thio)urea class of catalyst can find use in a host of potential applications, research and industrial environments.
- This article is part of the themed collection: Catalysis & biocatalysis in OBC


 Please wait while we load your content...
Please wait while we load your content...