Memristive plasticity in artificial electrical synapses via geometrically reconfigurable, gramicidin-doped biomembranes†
Abstract
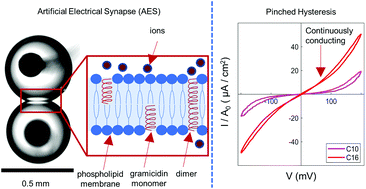
It is now known that mammalian brains leverage plasticity of both chemical and electrical synapses (ES) for collocating memory and processing. Unlike chemical synapses, ES join neurons via gap junction ion channels that permit fast, threshold-independent, and bidirectional ion transport. Like chemical synapses, ES exhibit activity-dependent plasticity, which modulates the ionic conductance between neurons and, thereby, enables adaptive synchronization of action potentials. Many types of adaptive computing devices that display discrete, threshold-dependent changes in conductance have been developed, yet far less effort has been devoted to emulating the continuously variable conductance and activity-dependent plasticity of ES. Here, we describe an artificial electrical synapse (AES) that exhibits voltage-dependent, analog changes in ionic conductance at biologically relevant voltages. AES plasticity is achieved at the nanoscale by linking dynamical geometrical changes of a host lipid bilayer to ion transport via gramicidin transmembrane ion channels. As a result, the AES uniquely mimics the composition, biophysical properties, bidirectional and threshold-independent ion transport, and plasticity of ES. Through experiments and modeling, we classify our AES as a volatile memristor, where the voltage-controlled conductance is governed by reversible changes in membrane geometry and gramicidin channel density. Simulations show that AES plasticity can adaptively synchronize Hodgkin–Huxley neurons. Finally, by modulating the molecular constituents of the AES, we show that the amplitude, direction, and speed of conductance changes can be tuned. This work motivates the development and integration of ES-inspired computing devices for achieving more capable neuromorphic hardware.


 Please wait while we load your content...
Please wait while we load your content...