Which is the most effective pristine graphene electrode for energy storage devices: aerogel or xerogel?†
Abstract
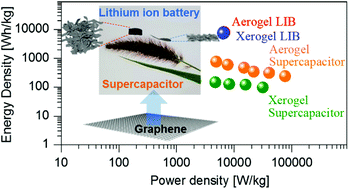
The morphological design of graphene materials is definitely important since their electrochemical properties as an electrode in energy storage devices are mainly dominated by their charge accessibility and active area. In this work, we present a systematic investigation on the prospects of a pristine graphene aerogel and a pristine graphene xerogel as electrode materials for both supercapacitors and lithium-ion batteries. We confirm that the graphene aerogel has a significantly higher surface area, needed for effective charge storage, than the xerogel, which offers a clear advantage for supercapacitors. In terms of battery performance, the quality of the pristine graphene raw materials is a more critical factor than their shape owing to the lithium intercalation mechanism. As a result, the graphene aerogel supercapacitors exhibited a specific capacitance of about 700 F g−1 at 10 mV s−1 in 1 M LiPF6 electrolyte, which is 3.6 times higher than the values for the xerogel devices. On the other hand, the electrochemical battery performances of the graphene aerogel and xerogel show no significant difference considering their high specific capacity of about 380 mA h g−1 at 1C. Further, the surface control kinetics of the graphene aerogel are much more dominant in the supercapacitor and battery applications than those of the xerogel. This study provides more practical approaches in order to design electrodes using lightweight, high-performance, and low-cost materials for the effective use of energy storage systems.
- This article is part of the themed collection: Editor’s Choice: 2D Materials for Energy Storage and Conversion


 Please wait while we load your content...
Please wait while we load your content...